Swab and Viral Transport Medium Market reflects a structural shift where procurement decisions are increasingly influenced by specimen stability, workflow compatibility, and supplier reliability rather than one-time volume spikes.
NEWARK, DE / ACCESS Newswire / May 5, 2026 / According to the latest market analysis by Future Market Insights, this growth is being driven by the sustained need for reliable specimen collection, standardized viral transport, and uninterrupted supply across diagnostic workflows. As healthcare systems transition from pandemic-driven surges to steady-state surveillance, swab and viral transport medium (VTM) products are no longer viewed as emergency-use consumables-they have become foundational to routine diagnostic infrastructure.
The market reflects a structural shift where procurement decisions are increasingly influenced by specimen stability, workflow compatibility, and supplier reliability rather than one-time volume spikes. Hospitals, diagnostic laboratories, and public health programs now rely on predictable replenishment cycles, making consistency in product performance and fulfillment a central requirement. In this environment, even minor variability in specimen collection or transport conditions can compromise diagnostic accuracy, delay results, and disrupt laboratory throughput-turning a basic consumable into a critical operational dependency.
Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.futuremarketinsights.com/reports/sample/rep-gb-11195
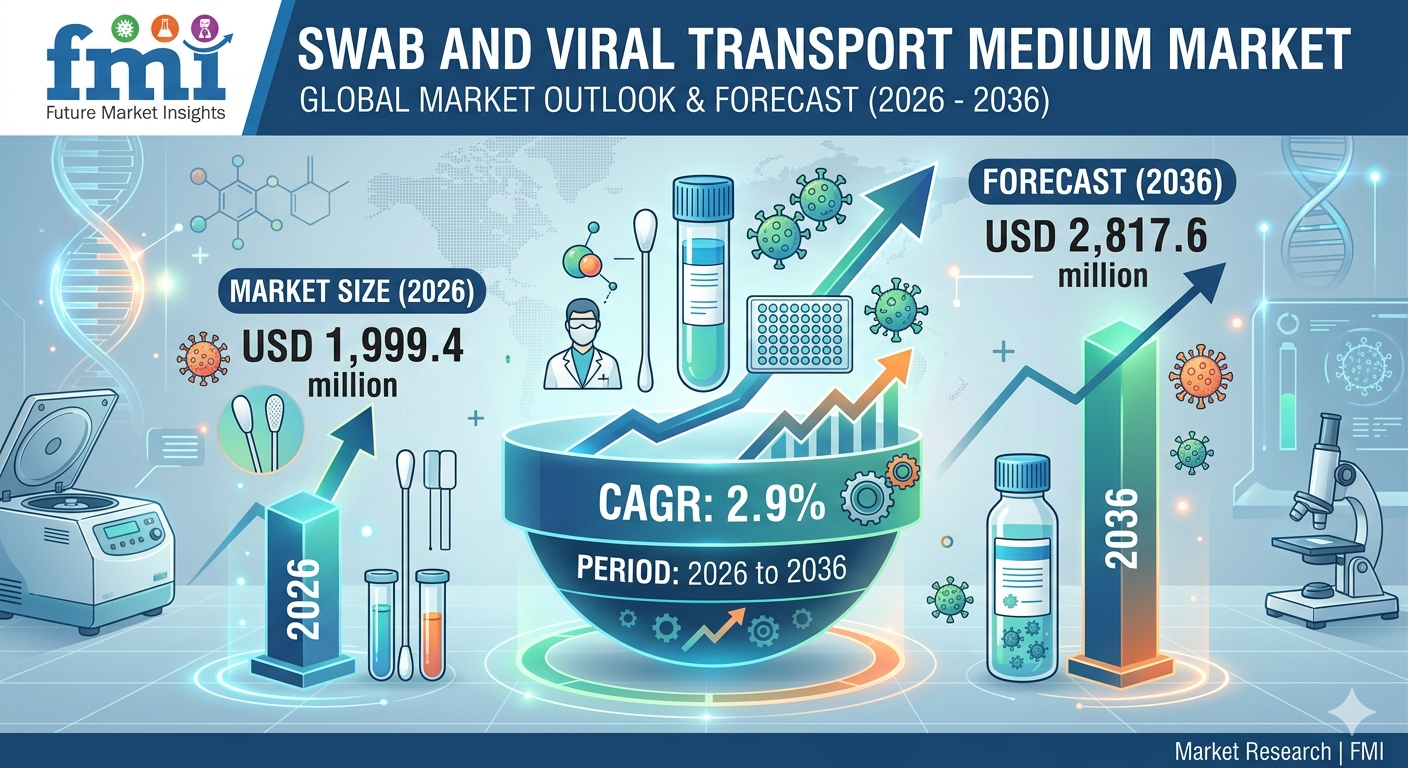
Quick Stats Snapshot (2026-2036)
Market size (2026): USD 1,999.4 million
Projected value (2036): USD 2,817.6 million
CAGR (2026-2036): 2.9%
Absolute dollar growth: USD 799.2 million
Leading product: Virus swabs (59.0% share)
Secondary product: Viral transport medium (41.0%)
Top application: Varicella-zoster virus (32.6%)
Fastest-growing markets: India (5.2%), China (4.2%)
Market Size and Forecast
The swab and viral transport medium market is projected to grow from USD 1,999.4 million in 2026 to USD 2,817.6 million by 2036, reflecting a steady CAGR of 2.9%. Unlike high-growth diagnostic technology segments, this category is characterized by stable, volume-driven expansion tied to routine testing needs rather than breakthrough innovation cycles.
Growth is anchored in recurring demand across hospitals, laboratories, and decentralized testing environments. As diagnostic systems normalize post-pandemic, procurement patterns are shifting toward long-term contracts, standardized SKUs, and cost-controlled replenishment models. This positions the market as a reliability-driven segment where operational continuity outweighs rapid expansion.
Growth Drivers: From Emergency Demand to Surveillance Stability
Public health surveillance programs: Year-round monitoring of respiratory viruses continues to drive baseline demand for specimen collection kits across healthcare systems.
Seasonal testing cycles: Influenza and respiratory syncytial virus (RSV) create predictable spikes, reinforcing recurring procurement patterns.
Decentralized testing expansion: The rise of clinics, outpatient centers, and point-of-care testing environments is increasing the need for standardized, easy-to-use specimen collection systems.
Workflow standardization: Laboratories increasingly prefer validated, consistent products that integrate seamlessly into established diagnostic protocols.
Market Challenges: Standardization Limits Pricing Power
Despite stable demand, the market faces structural constraints:
Commoditization pressure: Standardized product formats limit differentiation
Tender-driven pricing: Institutional procurement reduces pricing flexibility
Post-pandemic normalization: Reduced emergency demand impacts growth acceleration
Supplier switching barriers: Validation requirements slow adoption of new entrants
These challenges highlight a market where competition is less about innovation and more about execution, reliability, and compliance.
Opportunities: Where Competitive Advantage Is Built
Supply chain reliability: Vendors that ensure uninterrupted fulfillment gain long-term institutional trust.
Quality and compliance documentation: Products backed by validated performance claims and regulatory readiness strengthen supplier positioning.
Emerging market expansion: Countries such as India and China offer growth opportunities through expanding diagnostic infrastructure and preparedness-driven procurement.
Decentralized testing ecosystems: Standardized kits designed for distributed care settings are gaining traction as testing moves beyond centralized laboratories.
Segmentation Insights: Where Demand Is Concentrated
Product Type: Virus swabs dominate with 59.0% share, reflecting their essential role as the first step in specimen collection workflows. Viral transport media account for 41.0%, supporting preservation and transit.
Application: Varicella-zoster virus leads with 32.6% share, driven by consistent diagnostic demand linked to chickenpox and shingles testing. Influenza and RSV follow as recurring seasonal contributors.
End Users: Hospitals, diagnostic laboratories, and public health programs remain primary demand centers, with decentralized care settings emerging as a secondary growth layer.
Customize insights for your business strategy: https://www.futuremarketinsights.com/customization-available/rep-gb-11195
Regional Analysis: Growth Anchored in Emerging Markets
Asia-Pacific: The fastest-growing region, led by India (5.2% CAGR) and China (4.2%), driven by expanding diagnostic infrastructure and increased testing penetration.
North America: A mature market where demand is sustained through replacement cycles, established surveillance programs, and standardized procurement practices.
Europe: Characterized by compliance-driven procurement and stable demand, supported by structured public health systems in countries like Germany, France, and the United Kingdom.
Competitive Landscape: Execution Over Innovation
Key players include Becton, Dickinson and Company, Thermo Fisher Scientific, Cardinal Health, McKesson Corporation, and Quidel Corporation.
Competition is defined by:
Consistency in product performance
Ability to meet large-scale procurement demand
Regulatory compliance and documentation readiness
Distribution reach and fulfillment reliability
Companies with strong manufacturing infrastructure and established distribution networks maintain a structural advantage, particularly in institutional procurement environments.
Strategic Implications for Decision-Makers
Procurement leaders: Prioritize suppliers with proven reliability, validated product performance, and consistent supply capabilities.
Healthcare providers: Standardize specimen collection workflows to reduce variability and improve diagnostic accuracy.
Suppliers: Focus on operational excellence, compliance, and distribution scale rather than product differentiation alone.
Investors: Look for companies with strong institutional relationships and resilient supply chain capabilities.
Future Outlook: From Pandemic Spike to Baseline Necessity
Over the next decade, the swab and viral transport medium market will continue to evolve as a stable, infrastructure-driven segment within diagnostics. Growth will be defined not by sudden demand surges but by consistent, recurring use across surveillance programs and routine testing workflows.
The projected 2.9% CAGR reflects normalization-where these products transition fully into essential healthcare commodities underpinning global diagnostic systems.
Unlock 360° insights for strategic decision making and investment planning: https://www.futuremarketinsights.com/checkout/11195
Executive Takeaways
Demand is shifting from emergency-driven to routine surveillance-based procurement
Market growth is steady, supported by recurring testing needs
Product reliability and supply continuity are primary competitive factors
Emerging markets offer the strongest expansion opportunities
Institutional procurement dynamics favor established, compliant suppliers
In a diagnostic ecosystem where accuracy begins at the point of collection, swabs and viral transport systems have become indispensable. For healthcare systems and suppliers alike, consistency, reliability, and workflow integration are no longer advantages-they are baseline expectations.
Explore More Related Studies Published by FMI Research:
Hyperpigmentation Treatment Market: https://www.futuremarketinsights.com/reports/hyperpigmentation-treatment-market
Hypoventilation Management Market: https://www.futuremarketinsights.com/reports/hypoventilation-management-market
Keratitis Treatment Market: https://www.futuremarketinsights.com/reports/keratitis-treatment-market
Remote Healthcare Market: https://www.futuremarketinsights.com/reports/remote-healthcare-market
Tuberculous Meningitis Treatment Market: https://www.futuremarketinsights.com/reports/tuberculous-meningitis-treatment-market
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. Headquartered in Delaware, USA, with a global delivery center in India and offices in the UK and UAE, FMI delivers actionable insights to businesses across industries including automotive, technology, consumer products, manufacturing, energy, and chemicals.
An ESOMAR-certified research organization, FMI provides custom and syndicated market reports and consulting services, supporting both Fortune 1,000 companies and SMEs. Its team of 300+ experienced analysts ensures credible, data-driven insights to help clients navigate global markets and identify growth opportunities.
For Press & Corporate Inquiries
Rahul Singh
AVP - Marketing and Growth Strategy
Future Market Insights, Inc.
+91 8600020075
For Sales - sales@futuremarketinsights.com
For Media - Rahul.singh@futuremarketinsights.com
For web - https://www.futuremarketinsights.com/
For Web : https://www.factmr.com/
SOURCE: Future Market Insights, Inc.
View the original press release on ACCESS Newswire
